Home
Life Sciences
Life Sciences
Effective Regulatory Information Management
Compliance in Life Science industries is complex, and it’s only getting more difficult to maintain as companies globalize and introduce new technologies. Regulatory information management systems can help organizations remain up to date with regulations and effectively plan, execute and document compliance plans. As a distinguished Accruent Partner, Pentagon is well-positioned to offer the precise solution your industry requires.

Simplify compliance, streamline validation, and ensure quality and safety with Accruent’s highly secure, expertly validated SaaS EDMS.
Regulatory information management, as the name implies, is the process of managing all regulatory information. Historically, this information has been housed in a multitude of systems, including file shares, cloud systems, Excel documents and pen-and-paper repositories.
Today, this kind of disparate system is outdated and insufficient, particularly as organizations deal with challenges and burdens of compliance like:
- The increasing complexity of regulations and product portfolios
- Lack of transparency across systems and departments
- A global push for more collaboration and standardization
- Increased outsourcing of services and information
- High costs, including direct costs associated with planning, executing and documenting regulations.
- Indirect costs that come from time to market, operational costs, patient outcomes and product quality.
Meridian Asset Lifecycle Information Management Solution can simplify compliance and mitigate many of these burdens.
The Pentagon Advantage
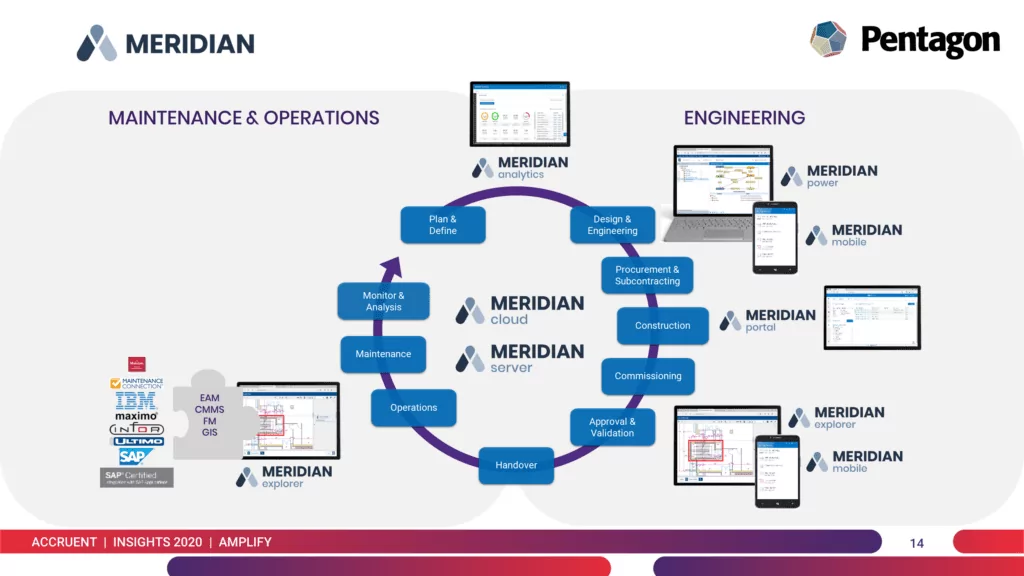
Introducing Meridian Asset Lifecycle Information Management Solution
Meridian Asset Lifecycle Information Management streamlines compliance and helps organizations effectively report compliance practices to regulatory agencies.
More specifically, this solution can help organizations in Life Sciences industries like pharmaceuticals, biologics and medical devices to:
- Prepare and submit product submissions in regulated formats.
- Manage product information, including compliance information.
- Effectively gather and submit information to regulatory agencies.
- Centralize all relevant product information.
- Organize authorizations and registrations.
- Track communication and correspondence for increased transparency.
Lead Your Industry With Meridian Cloud for Life Sciences
Propel your life sciences organisation to the Cloud, facilitate breakthrough innovation, and resolve your most pressing pain points with Meridian Cloud for Life Sciences.
Continuous compliance with 21 CFR Part 11, cGMP, GMP Annex 11, and other key regulations
Greatly Reduce the Cost and Effort of Validation
Deliver Up-to-Date High Quality Master Documents
Greater Collaboration with Contractors
At Pentagon Solutions, we’re more than just consultants; we’re your partners in progress. Let us help you conquer the evolving challenges in the life sciences industry and beyond. Contact us today to unlock new levels of efficiency and profitability.
HELP WITH
Digital Twins
Navigate the complexities of implementing digital twins, overcoming interoperability hurdles, and tackling integration challenges.
Change Management
Stay ahead of change with efficient digital workflows, enabling users to navigate pre-defined review and approval processes for seamless edits.
BIM Consultancy
BIM is a way for your business to reduce risk, cost, time, waste and energy from your project. Essentially BIM is going to save and make you money.
Asset Integration
Digital Construction integration focuses on improving productivity, reducing rework, and increasing project visibility and control.